Density of water in gcm32/2/2024  
Only three percent of the earth’s water is freshwater. It contains a concentration of dissolved salts less than 500 parts per million (ppm) of dissolved salts. What is Freshwater?įreshwater (or freshwater) is any naturally occurring water safe to drink. The specific gravity value of 0.2 means that 20% of the wooden cube will be submerged in water. It’s a ratio.įor something that floats, the specific gravity reveals what fraction of that object will be below the water while it’s floating.įor instance, consider a cube of wood floating on water with a specific gravity value of 0.2. It is the ratio of a material’s ρ with that of water at 4 ☌ (where it is most dense and is taken to have the value 999.974 kg m -3). Specific gravity (G) of a substance is its density divided by the ρwater. The ρ water is in the range of 0.9998 -0.999863 g/cm 3 at the same temperature and pressure The ρ ice is in the range 0.9167 -0.9168 g/cm 3 at 0 ☌ and a standard atmospheric pressure of 101,325 Pa This makes ice lighter than water, enabling it to float. Hence its ρ water decreases and becomes more than the initial volume. On cooling, It continues to contract and reduce in volume until 4☌.īeyond this temperature, its volume increases and becomes more than the initial volume. Due to this molecular structure, water behaves abnormally when cooled. To understand the concept of ice floating on water, we need to understand the structure of water.Įvery water molecule is made up of two hydrogen atoms bonded to one oxygen atom. However, this exceptional behavior is never seen in nature for any of these materials except water because melting points are very high except for water. Some materials like Silicon, Germanium, Bismuth, and water are less dense in solid form than in liquid form. Therefore, the ρ solid > ρ liquid > ρ gas The atoms or molecules of solids are more closers together than the atoms or molecules of the liquid. 
Generally, when a solid mass of a substance is put in a liquid of the same substance, it sinks. Since the ρ water of ice is less than the ρ water of water, therefore ice floats on water. Less dense matter floats on the dense liquid. The density of water (g/ml) = 8kg/(8 x10 -3 m 3 ) The mass (m) of 8 liters of water is 8 kg. Γ water = ρ water x g = 1000 kg/m 3 x 9.807 m/sec 2 = 9.807 kN/m 3 ( 1 N = kg⋅m/s 2) Density Of Water in g/ml-Example Problem Specific weight (γ) = (ρ x volume x g)/volume 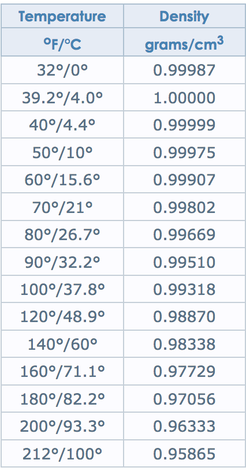 
The specific weight of water on Earth at 4☌ = 9.807 kN/m³ = 62.43 lbf/ft³. The specific weight of water refers to the weight per unit volume of a material. The density of fresh water on Earth at 4☌ is normally taken as 1000 kg/m 3. Common units of ρwater are grams per milliliter (1 g/ml), grams per cubic centimeter ( g/cm3), and pounds per cubic foot (lb/ft 3). The density of water ( ρ water )is around 1g/ml, but this value changes with temperature or impurities present in water. This is due to the large quantity of matter in iron than in wood.ĭensity (ρ) formula = mass (m) of substance ÷ volume (v) of substance The density of water in g/ml We will find that iron is heavier than wood. If we take equal volumes of wood and iron. It is the ratio of mass to volume of a substance. How much does a gallon of water weigh at its highest density?ĭensity is the quantity of matter per unit volume.The density of water in g/ml (effect of temperature).Density Of Water in g/ml-Example Problem.Calculating the specific weight of water. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
